Topics of Interest
Drug Shortages and Impact on Our Health Care System
By Sabrina Carrie, MD
Boston Children's Hospital
Harvard Medical School
In the last decade, drug shortages have been an ongoing health concern affecting millions of Americans. This issue is most certainly felt in the perioperative clinical setting where pain physicians continue to face significant limitations in drug supplies especially injectable, sterile and off patent medications. A steady worsening in the number of drugs in shortages has been observed from 2007 to 2011 and peaked in 2011 when approximately 267 were in short supply (See figure 1).
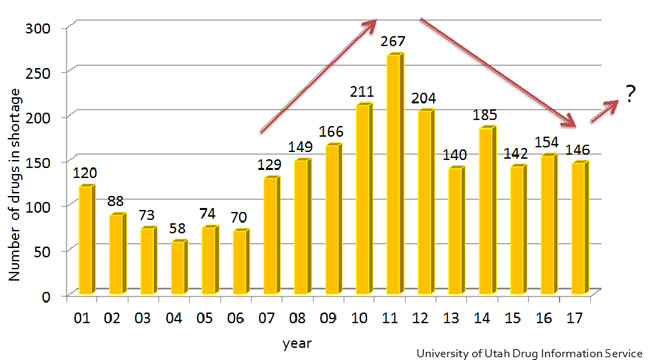
Figure 1. Trend in drug shortages
Since that time, there have been some improvements; however, the overall issue has remained a major crisis for the American health care system. Even today, in 2018, we are faced with significant drug shortages. Currently, the American Society of Health-System Pharmacists (ASHP) lists 156 drugs in short supply from a variety of drug classes ranging from antibiotics and chemotherapeutics, to cardiovascular drugs and hormonal therapies (See figure 2).
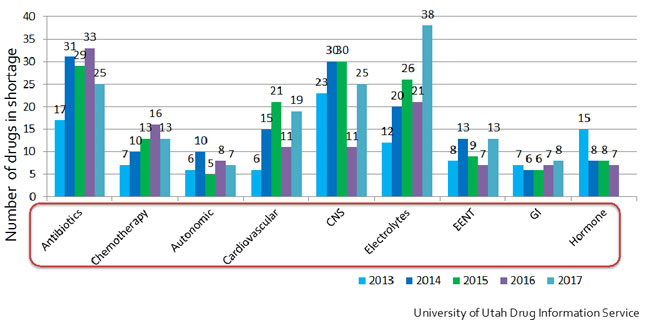
Figure 2. Trend in drug classes
When looking at the route of medication delivery, injectable forms are the most commonly affected representing about 72% of the total shortage followed by oral formulations at 15% (see figure 3).
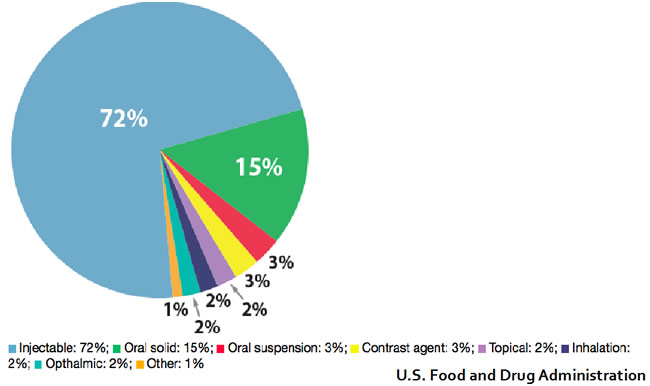
Figure 3. Route of delivery for drugs in shortage
More precisely, the products affecting perioperative care and pain management include opioids (morphine, hydromorphone, and fentanyl), ketamine, local anesthetics and intravenous (IV) fluids.
For the pediatric anesthesiologists and pain physicians, it is paramount to understand the factors affecting the hospital-based pharmaceutical demand and supply which dictates the availability of medications for our more vulnerable patient population. Furthermore, a better understanding of the issues surrounding drug shortages will allow clinicians to be advocates on a national level for better solutions to this health care crisis especially at a time when the focus is on the opioid crisis and overall overutilization.2 Lastly, awareness of the potential consequences of drug shortages can help us avoid the pitfalls and enable better prevention of patient safety concerns related to drug shortages.
The intricacies dictating the pharmaceutical-driven supply of medications has a clear and quite baffling disconnection to the hospital-based demand. On one hand, there is a relatively fixed demand for drugs such as opioids, ketamine, IV fluids as they are medically necessary for surgical, critical, emergency and anesthetics care. On the other hand, the supply of essential perioperative medications generated by the pharmaceutical companies remains relatively immune to the nationwide demand. One of the factors enabling this supply shortage originates from the simple fact that manufacturers have the monopoly. For instance, three companies; Pfizer, West-Ward and Fresenius are the major suppliers of IV opioids for the United States. Another contributing factor is the complex production process of these medications with specialized facilities and protocols including production quotas by the Drug Enforcement Administration (DEA) which further limits the access of smaller companies to the supply chain. There is also a lack of redundant manufacturing within each company with multiple drugs being manufactured on the same line of production. Therefore, manufacturing issues affecting one product line can easily affect multiple drugs in the same class and further worsen the shortage. Furthermore, from an economics standpoint, the reality is that investments in increased production capacity are not likely to occur in the foreseeable future. In fact, manufacturers of injectable, off patent medications operate with a tight profit margin with not much incentive to increase production. All of these factors, taken alone or collectively can lead to severe delays in response to market changes and manufacturing issues. Unfortunately, these numerous factors although seemingly straight forward, do not have an easy fix.
When looking more closely at the reported causes of drug shortages, it’s concerning that over 65% of inadequate supply directly results from product quality issues either because of facility remediation or product manufacturing issues (see figure 4).
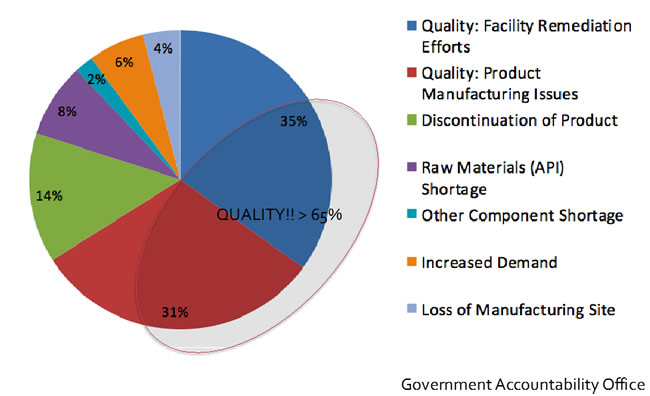
Figure 4. Causes of drug shortages
Manufacturing issues have included a range of causes from product line contamination with bacteria or fungus, crystallization and precipitation of residue, to equipment malfunction and natural disasters. For example, Hurricane Maria which hit Puerto Rico in September 2017 destabilized over 50 pharmaceutical facilities and is one of the major culprits for the current shortage. This natural disaster affected not only the displaced workforce, but also severely crippled the infrastructures and available electric power in the area. Baxter International, which owns ~40% of the IV fluid market in the United States, was also significantly affected in the process.
In the hospital day to day point of view, drug shortages have had a significant impact on the health care system. A 2010 survey of clinicians and pharmacists, revealed that most were frustrated and concerned by; the lack of advanced warning preceding a shortage, the uncertainty of the shortage duration and by the limited amount of resources invested into obtaining suitable alternatives and mitigating the negative effects caused by a shortage.4 One of the most concerning aspects of drug shortages that is often overlooked is patient safety concerns. In the same survey, more than 30% of respondents reported a near miss event, 25% an actual medication error and 20% an adverse patient outcome specifically related to a drug shortage. If only physicians were considered, 33% reported an adverse patient outcome directly related to the unavailability of a medication. Some of the reported medication errors around opioid shortages for instance involved the wrong dose of morphine or hydromorphone administered when an alternative concentration was available. Some of these events led to oversedation, others to the need for naloxone administration, ICU admissions and longer hospital length of stay. More importantly, there were two reported deaths when hydromorphone was given at the indented morphine dose.4 These reports from past shortages survey, while informative, are also worrisome and provide an impetus for clinicians especially pediatric anesthesiologists and pain physicians to stay vigilant while administering or prescribing alternate medications in the face of a drug shortages.
At the government level, there have been efforts to minimize these shortages. The Food and Drug Administration (FDA) is a federal agency that aims to protect and promote public health in a variety of arenas including assuring the availability of safe and effective drugs. They are in part responsible for the control and supervision of manufacturers and suppliers in the United States. They also oversee medications made abroad that are imported and eventually used for our patients locally. In 1999 they have created a subdivision, named the Drug Shortage Staff, to help prevent and alleviate or resolve drug shortages. In fact, they have been successful in many ways in reducing potentially catastrophic health care crisis. The FDA estimates for instance that in 2016 they have averted a total of 115 drug shortages, 87 of which were injectable drugs3. However, this task has not been easy given the constraints and particularities of the pharmaceutical market. This is especially true because they need to balance the need to regulate and set high quality standards for the production of medications. In fact, after the FDA required facilities to meet new quality standards through the FDA Modernization act, there was an overall reduction in industry capacities with many pharmaceutical companies eliminating generic injectable lines to save cost.
In October 2011, recognizing the need for policy change, an executive order from president Obama urged the FDA to use its authority to encourage early notification from manufacturers. This eventually led to the FDA Safety and Innovation act, which broadened the scope of early notification requirements. Unfortunately, the compliance with this legislation is marginal at best. About 40% of the time, manufacturers notify early regarding delays in production. Unfortunately, the majority of time, the FDA discovers manufacturing issues during inspections. (see Figure 5) Therefore, there is still a need for significant changes at a government level given the ongoing issues with the decrease of availability in essential perioperative medications.
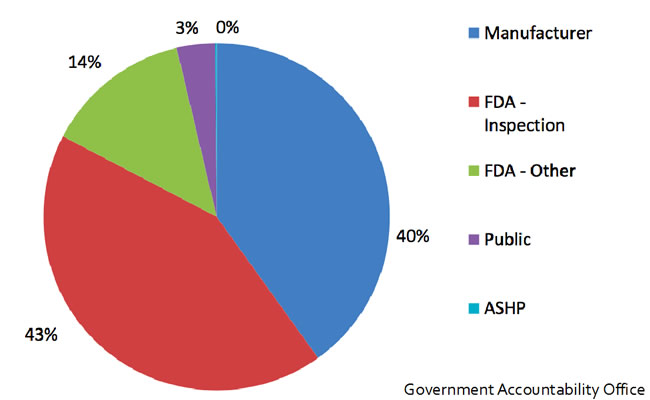
Figure 5. Delays in production: 43% of time, the FDA discovers manufacturing issues during inspections. About 40% of the time, manufacturers notify early regarding delays in production.
More recently, advocacy groups have urged the FDA for more effective solutions. In November 2017, a round table which included multiple national societies such as the ASHP and the American Society of Anesthesiology (ASA) met and drafted a letter to the FDA with suggestion to potentially expand the FDASIA. They suggested encouraging manufacturers with positive incentives to continue producing off-patent medications; they also urged the necessity for backup plans when delays in the supply chain are encountered.5 It is important to join these advocacy efforts as a Pediatric Pain Society collectively and as individual practitioners in order to be part of the leadership groups working towards a better healthcare system. In fact, we have a unique perspective as physicians treating the pediatric population. More importantly, pediatric anesthesiologist and pain treatment physicians need to remain attentive and strive to reduce errors and safety issues within their own practice, which may be catastrophic in this patient population.
References
- American Society of Health-System Pharmacists. https://www.ashp.org/drug-shortages/current-shortages
- The president’s commission on combating drug addiction and the opioid crisis. https://www.whitehouse.gov/sites/whitehouse.gov/files/images/Final_Report_Draft_11-1-2017.pdf
- https://www.fda.gov/Drugs/DrugSafety/DrugShortages/default.htm & https://www.fda.gov/Drugs/DrugSafety/DrugShortages/ucm441585.htm
- ISMP. ISMP survey on drug shortages. ISMP Medication Safety Alert! 2010;15(15):4
- Drug Shortages Roundtable: Minimizing Impact on Patient Care . Nov 2017
- Haninger et al. ASPE Economic Analysis of the Causes of Drug Shortages,2011
- Casey Moss. Hospitals are confronting a new opioid crisis: an alarming shortage of pain med. March 2018


