ASA Presentations
PLAY: A Phase 3 Study of Pharmacokinetics and Safety of Liposomal Bupivacaine for Pediatric Surgery
By Christopher F. Tirotta, MD, MBA;1 Alberto Jose de Armendi;2 Nicole D. Horn;3 Gregory B. Hammer;4 Michal Szczodry;5 Maria Matuszczak;6 Natalie Q. Wang;7 Richard Scranton;8 Robert Tracy Ballock9
1Nicklaus Children’s Hospital, Miami, FL; 2University of Oklahoma Health Sciences Center, Oklahoma City, OK; 3Indiana University School of Medicine, Indianapolis, IN; 4Stanford University, Stanford, CA; 5Shriners Hospital for Children, Chicago, IL; 6University of Texas, McGovern Medical School, Houston, TX; 7Pacira BioSciences, Inc., Parsippany, NJ; 8Lyndra Therapeutics, Watertown, MA; 9Cleveland Clinic, Cleveland, OH
INTRODUCTION
Liposomal bupivacaine (LB) is a long-acting formulation of bupivacaine approved for prolonged postsurgical analgesia in adults via local infiltration or interscalene brachial plexus nerve block. Observational data suggest that LB may be safe and effective in pediatric patients, but the pharmacokinetics (PK) and safety of LB in pediatric patients have yet to be described in controlled clinical trials.
METHODS
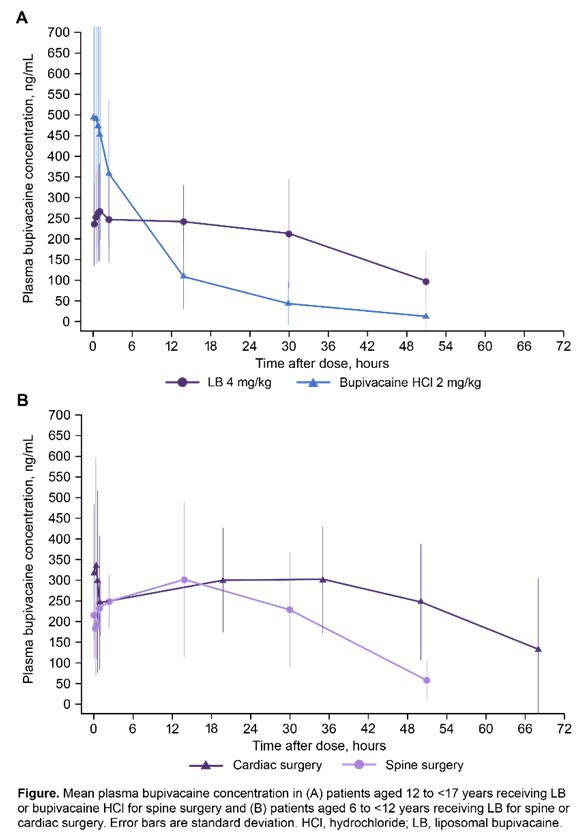
A multicenter, open-label, phase 3 trial (PLAY; NCT03682302) evaluated the PK and safety of LB in pediatric patients aged 6 to <17 years. Each site obtained institutional review board approval. Patients were enrolled into 2 groups: patients aged 12 to <17 years receiving LB 4 mg/kg or bupivacaine hydrochloride (BUPI HCl) 2 mg/kg for spine surgery (group 1) or patients aged 6 to <12 years receiving LB 4 mg/kg for spine or cardiac surgery (group 2). The maximum total doses of LB and BUPI HCl were 266 and 175 mg, respectively, administered at the end of surgery via local infiltration. Use of perioperative opioids and nonsteroidal anti-inflammatory drugs was permitted in accordance with each site’s standard of care. The primary and secondary objectives were to evaluate the PK and safety of LB, respectively. PK end points included area under the plasma bupivacaine concentration-time curve (AUC), maximum plasma concentration (Cmax), time to Cmax (tmax), and apparent elimination half-life (t1/2) assessed by noncompartmental analysis. Adverse events (AEs) were monitored until day 30 after surgery.
RESULTS
Baseline characteristics were comparable across treatment arms in group 1. Patients in group 2 generally had a higher American Society of Anesthesiologists physical status class than group 1. Patients in group 1 treated with LB had lower geometric mean Cmax versus patients treated with BUPI HCl (337 vs 488 ng/mL; Figure). Mean AUC0-∞ was greater in patients receiving LB versus BUPI HCl (12,257 vs 5064 >ng⋅h/mL). Median tmax in group 1 was similar between LB and BUPI HCl (1.1 and 0.9 h, respectively); however, LB also resulted in a second tmax (median 18.0 h). Geometric mean t1/2 was longer in patients receiving LB versus BUPI HCl (21.2 vs 7.4 h). In group 2, mean Cmax after administration of LB was lower in patients undergoing spine versus cardiac surgery (320 vs 447 ng/mL), and a second tmax was observed for both spine and cardiac surgery (median tmax of 15.3 and 30.1 h, respectively). Individual Cmax values after LB administration remained well below the toxic threshold (~2000 ng/mL) in all individuals in both age groups. In group 1, the incidence of treatment-emergent AEs (TEAEs) was comparable between LB and BUPI HCl (61.3% [n=31] and 73.3% [n=30], respectively). There were few neurologic or cardiac TEAEs in group 1 with LB (2 [6.5%]) and BUPI HCl (3 [10.0%]); none were considered treatment related. In group 2, 100% and 31% of patients undergoing spine (n=5) and cardiac (n=9) surgery experienced TEAEs, respectively, with no neurologic or cardiac TEAEs. Across all patients, TEAEs were mild or moderate in severity, except for 1 severe TEAE with BUPI HCl (constipation). There were no discontinuations due to TEAEs or deaths.
DISCUSSION
Plasma bupivacaine levels following local infiltration with LB in pediatric patients remained below toxic thresholds after both spine and cardiac surgery. All AEs observed with LB were mild to moderate in severity, and the incidence of neurologic and cardiac AEs was low. These data support the safety of LB 4 mg/kg administered via local infiltration in pediatric patients for spine or cardiac surgery.
References
- Chughtai M, Sultan AA, Hudson B, et al. Liposomal Bupivacaine Is Both Safe and Effective in Controlling Postoperative Pain After Spinal Surgery in Children: A Controlled Cohort Study. Clin Spine Surg. 2020.
- Cohen B, Glosser L, Saab R, et al. Incidence of adverse events attributable to bupivacaine liposome injectable suspension or plain bupivacaine for postoperative pain in pediatric surgical patients: A retrospective matched cohort analysis. Paediatr Anaesth. 2019;29(2):169-174.
- Day KM, Nair NM, Griner D, Sargent LA. Extended Release Liposomal Bupivacaine Injection (Exparel) for Early Postoperative Pain Control Following Pharyngoplasty. J Craniofac Surg. 2018;29(3):726-730.




